Bahrain
Bahrain approves Sinopharm COVID-19 vaccine for ages 3-17
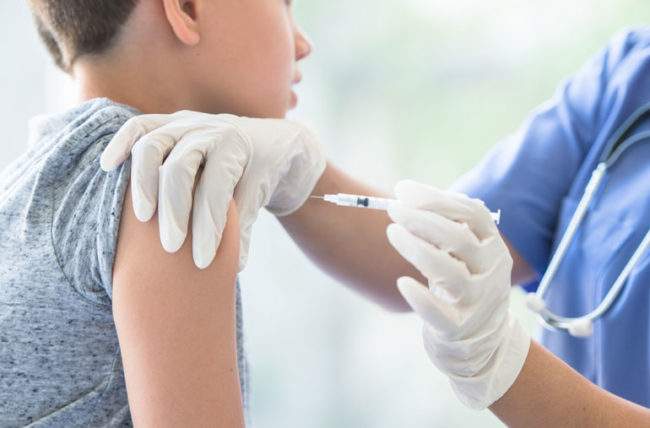
The National Medical Taskforce for Combatting the Coronavirus (COVID-19), today announced the approval of the vaccination of children aged 3-11 suffering from comorbidities and low immunity with the Sinopharm vaccine.
The Taskforce further noted the approval of the Sinopharm vaccine for ages 12 to 17 years, in addition to the Pfizer-BioNTech vaccine.
The Taskforce stressed the importance of ensuring eligible children receive the vaccine to safeguard public health, adding that the incubation period for the virus in this age group without symptoms extends up to 4 days, which may increase the spread of the virus.
The Taskforce highlighted that vaccination registration for ages 3-17 will be available on the MOH website and via the BeAware app.




